|
For example, Copper (Cu) and Oxygen (O2) combine to form Copper oxide (CuO2).Ĭu + O2 → CuO2 Disproportionation reaction: For example, H2 and O combine to form H2O, but if it can be broken down into H2 and O.Ī combination reaction, also named a synthesis reaction, is the combination of two compounds to form a single compound or a molecule. 
It is the reverse of a combination reaction where the breakdown of molecules occurs. In a decomposition reaction, a molecule is broken down to form elements. 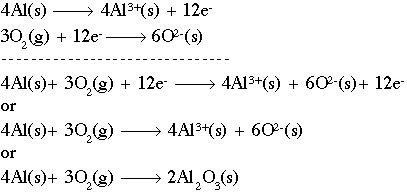
Here are some types of redox reaction: Decomposition Reaction: Related: To balance the reaction you can use our balancing chemical equations solver. The copper oxide and hydrogen ions combine to form copper and a water molecule. The reduction reaction for chlorine (Cl) is: The oxidation reaction for Hydrogen (H) is: What will the Reaction between Copper oxide and Hydrogen produce? The hydrogen and chlorine ions combine to form hydrogen chloride. The reduction reaction for Chlorine (Cl) is: The oxidation reaction for hydrogen (H) is: So, this reaction is between hydrogen and chlorine where the hydrogen is oxidized, and the chlorine is reduced. In the redox reaction, one atom is oxidized while the other is reduced. What will the Redox Reaction between Hydrogen and Chlorine produce? Therefore, We will give you some of the examples: Example 1: As redox reaction calculator helps you whenever you need it but you have to do it manually sometimes. Some examples of redox reactions are given to help you understand it better. CuO is an oxidizing agent because it oxidizes H2O while H2 is a reducing agent because it reduces CuO. On the other hand Hydrogen atom (H2) gains an oxygen atom making an H2O molecule so it is oxidized, which is found by determining the oxidation number. In the above chemical reaction Copper Oxide (CuO) loses an oxygen atom so that it is reduced. In short, the exchange of electrons occurs where one substance loses an electron and one substance gains an electron. In a redox reaction, chemical species transfer the electrons, leading to changes in those chemical species' oxidation state. Redox reactions, also known as oxidation-reduction reaction, is an important concept in the field of chemistry. You just input your query and our calculator will solve it giving you a step-by-step solution. The need to manually calculate the reactions is gone, rather our calculator will help you. This is an online tool that automatically calculates the stoichiometry of reactions and balances the reduction reaction’s equations making it easier for everyone who uses it. The redox equation calculator is here to make the process of determining redox reactions easy. Post memes/jokes in /r/chemistrymemes and /r/chemistryjokes.Introduction to Redox Reaction Calculator: Any such posts will be deleted.Īsk education and jobs questions in the current weekly topic. 
If you're looking for a more concentrated, advanced discussion of chemistry topics among professionals and grad students, check out /r/Chempros.īefore asking "What chemical is this?" see this chart. Click here for the OSHA chemical data site and here for a multicompany MSDS aggregate search. If you spill/injure yourself contact medical professionals and read the MSDS, do not post to this reddit. Yes links to blogs, images, videos, comics, and infographics are okay especially if they are on your personal website. No physorg, sciencedaily, or other press release aggregator spam! 
If a caption or explanation is included this helps, but please use your discretion.īefore asking about chemical drawing/illustration programs, look at your school's IT/software website and see if they provide an institutional license of ChemDraw (hint: if they have a chemistry department, they will) Likewise, simple pictures of uninteresting and garden variety chemistry-related things are not appreciated. No memes, rage comics, image macros, reaction gifs, or other "zero-content" material. However, academic discussions on pharmaceutical chemistry and the science of explosives are permitted. Rules: Violating a rule will result in a ban.Īsk homework, exam, lab, and other undergraduate-level questions at ChemicalForums otherwise it will be deleted.ĭiscussions on illicit drug synthesis, bomb making, and other illegal activities are not allowed and will lead to a ban.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
